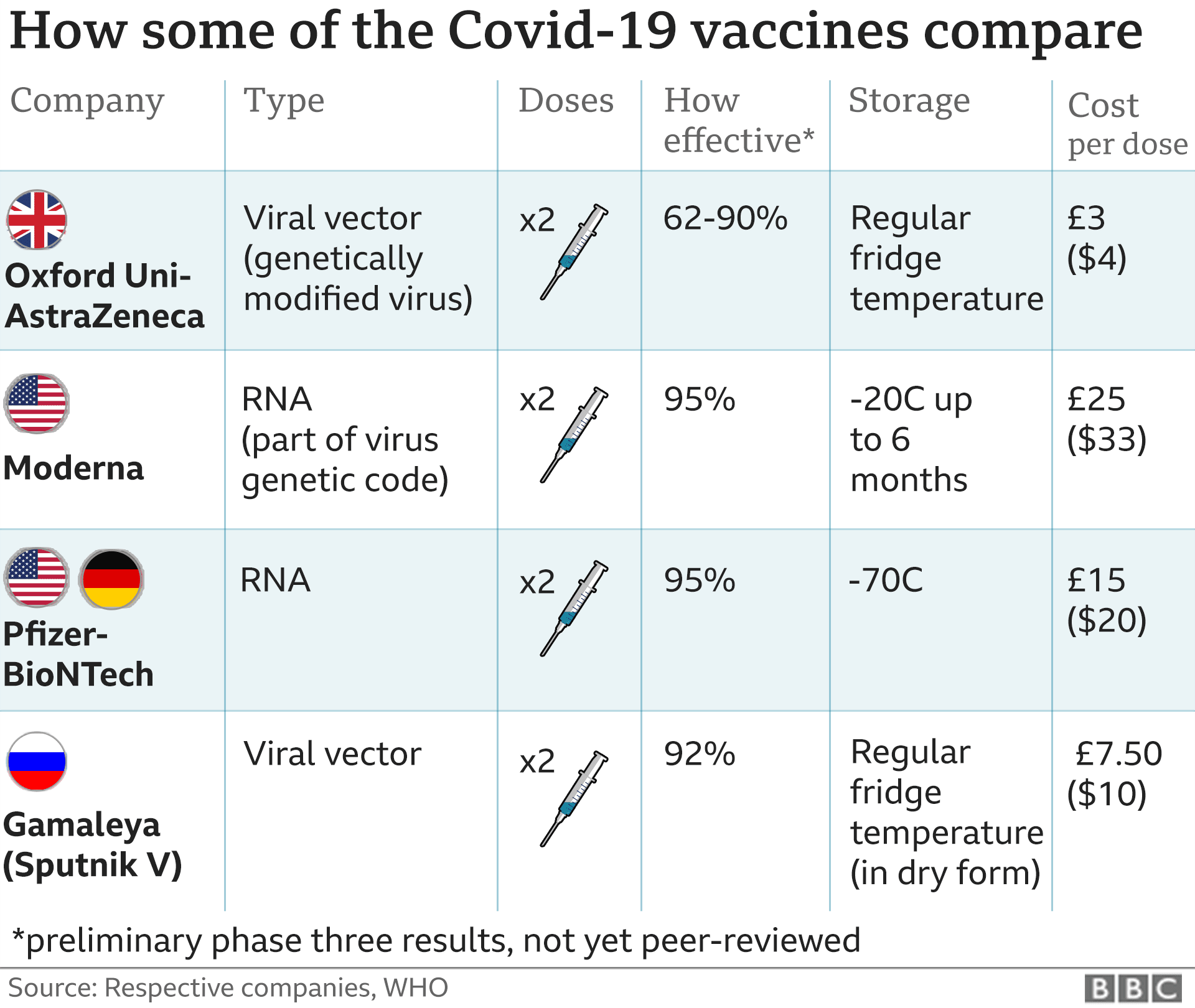
None of the children had previously been diagnosed with COVID-19. A week after the second dose was given, there were no cases of COVID-19 in the 1,005 children given the Pfizer-BioNTech vaccine.Īmong 978 children given the placebo, there were 16 cases of COVID-19. The other children were given a placebo shot. Of this group, about half were given the Pfizer-BioNTech COVID-19 vaccine. The results suggest that the vaccine is about 91% effective in preventing COVID-19 in this age group.įor children ages 12 through 15, the FDA reviewed a vaccine study of more than 2,200 U.S. Among 663 children given the placebo, there were 16 cases of COVID-19. Among 1,305 children given the vaccine, there were three cases of COVID-19. None of the children in this analysis had been previously diagnosed with COVID-19. The FDA also took an early look at cases of COVID-19 that occurred one week after children were given a second dose of the vaccine. Also, if your child recently had COVID-19, you may talk to a health care professional about delaying vaccination by up to three months. A longer interval might give more protection against COVID-19 and lower the risk of rare heart problems, such as myocarditis and pericarditis. The CDC recommends that people who get a Novavax vaccine series also get one shot of either Moderna or Pfizer-BioNTech COVID-19 bivalent vaccine at least eight weeks after their second Novavax shot.Īn eight-week interval between the first and second doses might be best for some people, especially males ages 12 to 39. The second shot is recommended 3 to 8 weeks after the first shot. For people age 12 and older the series is two shots of the vaccine. This can be given at least eight weeks after the first shot. For people in this age group who had one shot of the monovalent vaccine, an additional dose of the bivalent vaccine is recommended. That shot can be given at least eight weeks after the second monovalent vaccine.įor people age 6 and older one shot will bring them up to date. After the first dose, the second is given at least 4 to 8 weeks later.įor kids between ages 6 months and 5 years who previously got two shots of the monovalent Moderna COVID-19 vaccine, an additional dose of the currently available bivalent vaccine is recommended. For children between ages 6 months and 5 years, the series is two doses. 
Those who already had one dose of monovalent vaccine can get a bivalent shot at least eight weeks after the original dose.įor people age 5 and older who have already had one dose of the bivalent vaccine, the CDC hasn't recommended more shots. People age 5 years and older who are unvaccinated need one shot to be considered up to date. If a child has had three shots of the monovalent Pfizer-BioNTech COVID-19 vaccine, the child is eligible for one additional dose of the bivalent vaccine at least eight weeks after their last monovalent shot. The third shot is given at least 8 weeks after that. 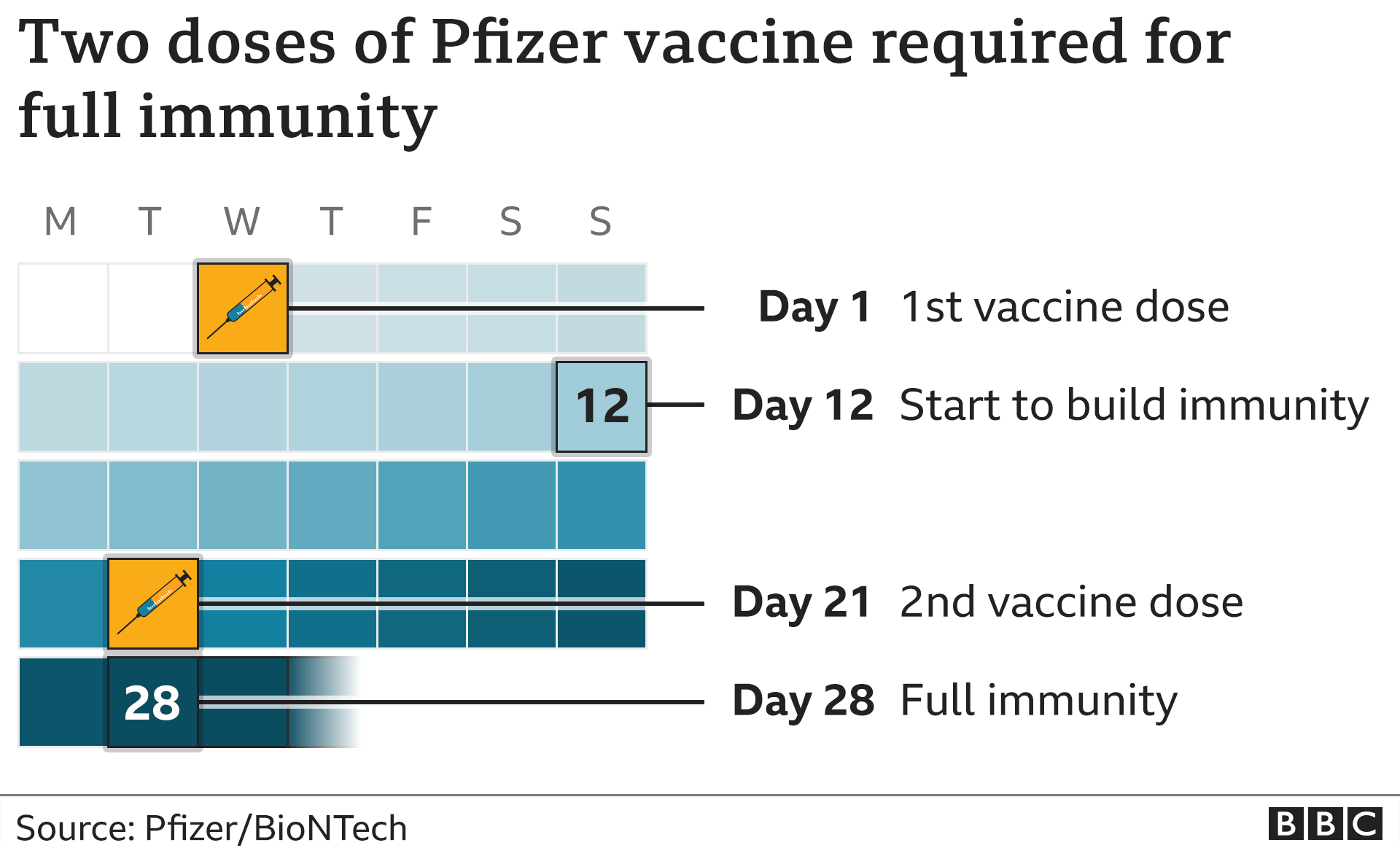
"That's why you tend to have more strong side effects from that initial vaccine.After the first dose, the second shot is given at least 3 to 8 weeks later. "If you were ever to be exposed to the infection again, your body would basically be able to respond quickly and more robustly that second time around," he added. Vivek Cherian, an internal-medicine physician in Baltimore, told Insider. "If you've already had a COVID-19 infection, you've developed memory cells from that infection," Dr. About 73% of vaccine recipients who'd previously had COVID-19 developed side effects after dose one of Pfizer-BioNTech's or Moderna's shot, compared with 66% of vaccine recipients who'd never gotten infected. People who've had COVID-19 may develop more side effects after dose 1Ī small study from the Icahn School of Medicine at Mount Sinai found that vaccine side effects such as fatigue, headaches, and chills were more common among people with preexisting immunity to the coronavirus than people who'd never been infected. Fevers were also far more common among second-dose recipients than first-dose recipients in both trials. In Moderna's trial, meanwhile, about five times as many participants developed chills after their second dose as did after their first. Roughly twice as many participants in Pfizer's trial developed chills and joint pain after their second dose than after their first. Account icon An icon in the shape of a person's head and shoulders.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
